Magic Traveling Flame
You may think it’s impossible or that we have completely lost it, but we are going to show you a simple trick that will surprise […]

There’s a lot of chemistry behind the simple lighting or extinguishing of a flame. Would you believe us if we told you that you could extinguish a flame just by placing it partway into a graduated cylinder? What if we told you that you could relight the fire without a match or lighter? Both propositions seem unlikely, if not downright impossible. With the Flame Light Relight experiment, however, you’ll see how a few household items can mix together and create a fire-based experience you have to see to believe.

Add 1 teaspoon of baking soda to each of the two graduated cylinders.

Add 1 teaspoon of yeast to both each of the graduated cylinders.

Shake, spin, and twirl the graduated cylinders to mix up the baking soda and yeast combination.

Add a generous splash of hydrogen peroxide to one of the graduated cylinders.

In the other graduated cylinder, pour in a generous amount of white vinegar.
The addition of hydrogen peroxide and vinegar to their respective cylinders creates a reaction with bubbling and fizzing in each of the cylinders. This is how you know you’re on the right track.
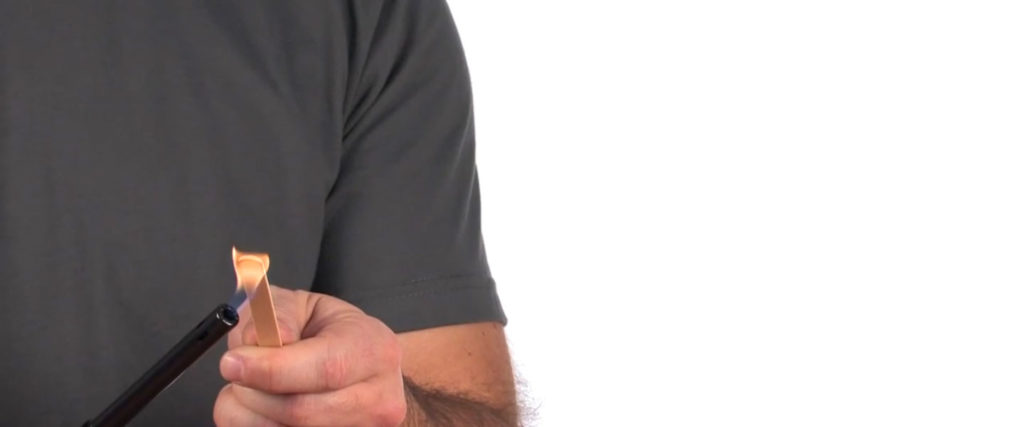
Use a lighter to ignite a popsicle stick. Make sure that you have a strong, consistent flame on the popsicle stick before continuing.

Stick the lit end of the popsicle stick down into the graduated cylinder that contains vinegar. You do not need to touch the bubbles or liquid. The flame will extinguish.

Now, move the still-smoking popsicle stick into the cylinder that contains hydrogen peroxide. Again, no need to touch the bubbles or liquid in the cylinder. The ember where a flame once was will begin to glow more intensely as it stays in the cylinder until the flame relights!

Repeat placing the popsicle stick into the vinegar cylinder and the flame will once again extinguish. Replace the popsicle stick in the hydrogen peroxide cylinder and, once again, the flame is reignited.
Most flames require oxygen, fuel, and sufficient heat to ignite and stay lit. These three components of fire are referred to as the fire triangle or combustion triangle. Removal of any of the three components will cause the flame to extinguish or “go out.”
In the instance of the popsicle flame, the three necessary components are present during the initial lighting of the fire. The heat generates from a separate flame, the lighter. The wood of the popsicle stick provides the fuel. Finally, the oxygen level present in the atmosphere is enough to sustain a flame.
When you stick the flaming popsicle stick into the first graduated cylinder it extinguishes. That means one of the three components of the fire triangle is missing, but which one?
The secret lies in the bubbling mixture in the cylinder. The baking soda (also known as sodium bicarbonate) is a base. The vinegar, or acetic acid, is a weak acid. When baking soda and vinegar are combined, the immediate acid-base reaction creates carbonic acid. Carbonic acid is unstable and decomposes into carbon dioxide (CO2) and water (H2O). The bubbling that you see inside of the cylinder is the production of the CO2 gas. When you dip the popsicle stick into the cylinder, you’re exposing the flame to concentrated CO2 gas. The lack of oxygen extinguishes the flame.
Upon placing the popsicle stick into the second graduated cylinder, the ember begins to glow more intensely until the flame reignites. The invisible property at work is the reintroduction of oxygen (O2). The high concentration of O2 in the cylinder makes the heat source more intense until the flame ignites again.
Hydrogen peroxide (H2O2) is fairly unstable and always trying to decompose into water (H2O) and oxygen (O2). When yeast is mixed with hydrogen peroxide, it acts as a catalyst to the decomposition. This creates oxygen at a much faster rate which you can see bubbling inside of the graduated cylinder. Placing the partially glowing ember into the concentrated oxygen completes the fire triangle and reignites the flame.
Igniting and extinguishing a flame in a cylinder is cool, but it isn’t a science fair project. You can create a science fair project by identifying a variable, or something that changes, in this experiment. Let’s take a look at some of the variable options that might work:
That’s just one idea, but you aren’t limited to that! Try coming up with different ideas of variables and give them a try. Remember, you can only change one thing at a time.